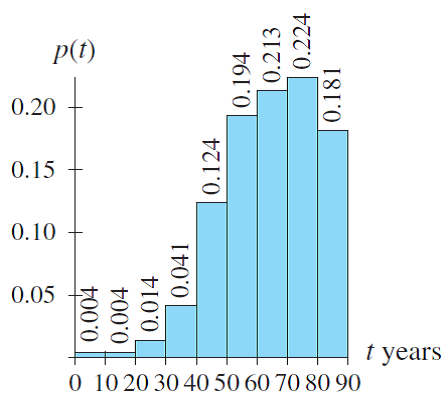 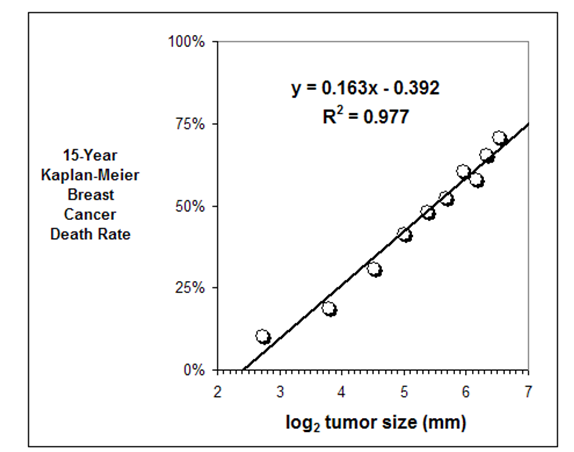
The following considerations apply when selecting the most appropriate health guidelines and cancer risk values: However, it is important for health assessors to understand ATSDR’s selection process. EPA develops IURs as a result of quantitative evaluation of inhalation exposure to a suspected carcinogenic contaminant.Īgain, remember that PHAST will select the most appropriate health guidelines to evaluate non-cancer effects and cancer risk values to evaluate cancer effects for your scenarios.Measure of the relative potency of various carcinogens from inhalation exposures.Are estimates of increases in cancer cases in a human population.EPA develops CSFs as a result of quantitative evaluation of oral exposure to a suspected carcinogenic contaminant.Measure of the relative potency of various carcinogens from oral exposures.Are estimates of daily inhalation exposures to a contaminant that are likely to be without a discernible risk of deleterious effects to the general human population, including sensitive subgroups, during a lifetime of exposure.Ĭommon Cancer Risk Values Used by ATSDR EPA-Derived Cancer Risk Values.Are estimates of daily oral exposures to a contaminant that are likely to be without a discernible risk of deleterious effects to the general human population, including sensitive subgroups, during a lifetime of exposure.ĮPA-Derived Reference Concentrations (RfCs).Are available for oral and inhalation exposures. 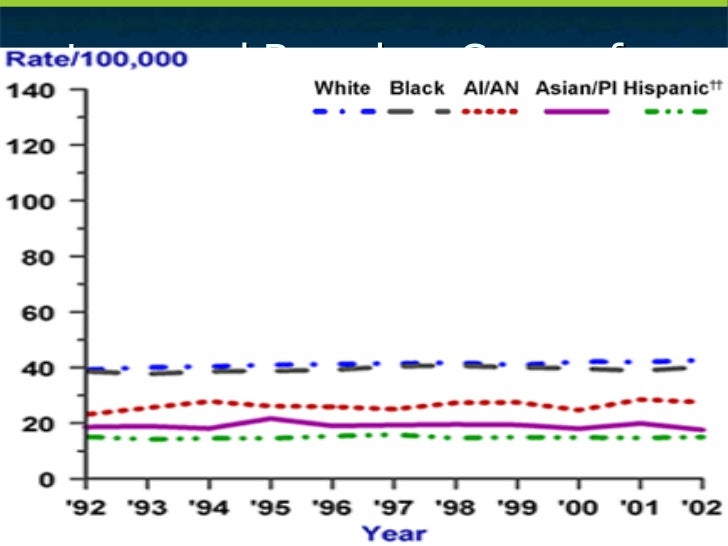
Are derived for acute (1-14 days), intermediate (15-364 days), and chronic (365 days and longer) exposure durations.Are set below levels that might cause adverse health effects in most people, including sensitive populations.Represent estimates of the daily human exposure to a contaminant that, based on ATSDR evaluations, are not expected to cause non-cancer health effects during a specified exposure duration.Common Health Guidelines (Non-Cancer) Used by ATSDRĬommon Health Guidelines Health GuidelinesĪTSDR-Developed Minimal Risk Levels (MRLs) The values do not represent thresholds of toxicity. Health guidelines (used to evaluate non-cancer health effects) ( see descriptions in this table) and cancer risk values (used to estimate cancer risks) ( see descriptions in this table) are derived from data in the epidemiologic and toxicologic literature with appropriate uncertainty or safety factors applied to ensure they are set at levels below those that could result in harmful health effects. Different health guidelines are available for exposure routes (ingestion, inhalation), exposure durations (acute, subchronic/intermediate, and chronic), and health endpoints (carcinogenic, non-carcinogenic). Important: PHAST will make this selection for you, but it is important for health assessors to understand ATSDR’s selection process. Selecting Appropriate Health Guidelines and Cancer Risk Valuesīefore performing the HQ and CR calculations, you will need to determine the most appropriate health guidelines or cancer risk values, respectively, to use for evaluating your site-specific exposure doses or adjusted air concentrations. Once you have your estimated exposure doses and adjusted air concentrations, you will use health guidelines and cancer risk values to estimate the potential non-cancer health effects and cancer risks, respectively, for each contaminant and exposure pathway.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
